Structural Properties of Prokaryotic GTPases
GTPases, or G-proteins, are one of the largest and most functionally diverse groups of proteins represented in genomes. They are responsible for regulating numerous cellular processes such as protein synthesis, cell growth and nuclear transport. GTPases are called “molecular switches” based on their ability to interconvert between a guanosine 5'-diphosphate (GDP)- and guanosine 5’-triphosphate (GTP)-bound state. Cycling between these states causes alterations in the surface properties of the GTPase promoting periodic interactions with a cognate partner in a cellular pathway. All GTPases have the same basic protein fold and mechanism of action but they vary greatly in their sequence, size and shape. The GTPase core contains a six-stranded ß sheet surrounded by 5 a helices. The nucleotide-binding site of the protein is defined by four conserved amino acid sequence motifs, termed the G-1 through G-4 boxes, which are located within flexible regions of the GTPase core. The conformational changes associated with the switching mechanism can be localized to two areas of the GTPase core known as the switch I and II regions. The GTPase cycle, consisting of nucleotide binding, hydrolysis and release, is unique for each individual GTPase both in terms of timing and also in the magnitude of the conformational changes that take place.
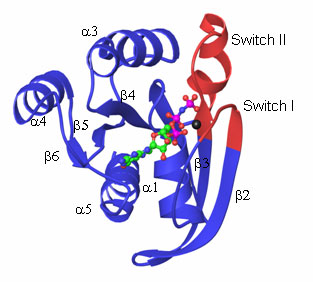
The GTPase Structural Core. Ribbon diagram of RAS, the archetypical GTPase, with bound GTP and Mg2+ shown in ball-and-stick model. The Switch I and II regions are colored red. (PDB 121P)
The Classic GTPase Cycle
GTPases exist in two major states, a GTP- and GDP-bound form. In most systems, the GTP-bound form of the GTPase is the “on” state implying that it is able to associate with cognate partners. Transition to the GDP-bound form or “off” state occurs through hydrolysis of GTP to GDP. This slow reaction can be stimulated by a GTPase-activating protein or GAP. The protein is restored to the active state through dissociation of GDP and binding of GTP. Similar to the hydrolysis event, this exchange is slow and therefore is facilitated in some systems by a GEF or guanine-nucleotide exchange factor.
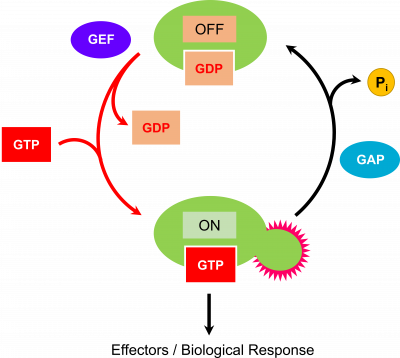
Biochemical Properties of Prokaryotic GTPases
The biochemical traits we generally associate with GTPases have been compiled from studies focused on the activities of eukaryotic G-proteins. Conversely, few details are known about how bacterial GTPases function within bacteria aside from the translation factors EF-G and EF-Tu, and the signal recognition particle and its receptor, Ffh and FtsY. All prokaryotic GTPases have activities that are essential to fundamental cellular process such as viability, protein synthesis and pathogenesis. As such they are attractive targets for antimicrobial development. Strikingly, those characterized thus far regulate some aspect of ribosome function or assembly, the majority binding directly to the ribosome. This association, in turn, influences the catalytic cycle of the GTPase.
We know little about how the molecular mechanisms of action of many of these GTPases dictate their cellular function. Therefore, my group is using a combination of structural biology, biochemistry and microbiology to elucidate the molecular mechanism of action of several families of unique bacterial GTPases and relate these properties to their activities within the bacteria. Long term, unique characteristics of each protein will be targeted for antimicrobial agent development.
BipA
BipA is a highly conserved prokaryotic GTPase which functions as a regulator of virulence processes in many strains of globally problematic bacteria. In Escherichia coli and Salmonella typhimurium pathogens, BipA has been implicated in controlling bacterial motility, enhancing attachment and effacement (A/E) processes, upregulating the expression of certain virulence genes and thwarting host defense mechanisms. BipA is required in E. coli and Bacillus subtilis for low temperature growth and can be linked to ppGpp levels in B. subtilis, suggesting that BipA is somehow involved in stress responses in bacteria. Despite these findings, the exact cellular function of BipA remains elusive. We demonstrated that BipA forms two distinct biologically relevant complexes with the ribosome, 70S:BipA and 30S:BipA. The formation of these species is dependent upon the guanine nucleotide pool in the cell, specifically GTP and ppGpp, an alarmone responsible for adaptation to altered growth conditions in bacteria. We hypothesize that GTP and ppGpp compete for binding to BipA and induce conformational changes that promote the differential association of BipA and the ribosome. BipA would therefore act as an intermediary between the ribosome and the cellular environment and its structural and regulatory properties influence how the bacteria respond to environmental conditions. By determining BipA's molecular mechanism of actions, we will better understand stress and virulence processes in bacteria.
Previous Projects
EngA
EngA is unique both among eukaryotic and prokaryotic GTPases by virtue of its domain architecture. EngA orthologues, present in all bacterial genomes and Arabidopsis, are the only known proteins to contain two tandemly repeated GTP-binding domains (GD1 and GD2) connected by a variable length acidic linker. This distinct domain architecture leads us to speculate that there are multiple levels of inter- and intra-domain regulation occurring within the protein and between this protein and its target partners. Recent evidence indicates that EngA is involved in 50S ribosomal subunit assembly in bacteria and that cooperative activity of both GTPase domains are required for this function. Using biochemical, biophysical and structural methodologies we are beginning to uncover how intra- and intermolecular interactions involving S. typhimurium EngA contribute to its molecular mechanism of action. As EngA is an essential bacterial protein universally conserved in pathogenic bacteria, the long-term goal of these studies is to develop inhibitors of EngA, which have potential as antimicrobial therapeutics.
Nucleostemin
Nucleostemin (NS) is a novel nucleolar GTPase preferentially expressed in the nucleoli of neural stem cells, embryonic stem cells and several cancer cell lines . Recent studies have shown that NS is required for stem cell and cancer cell maintenance, and that NS modulates proliferation of these cells. Although it is not well understood, GTP-driven NS localization to the nucleolus is key to the functioning of the protein. Deletion studies have identified several functional domains in the protein. These include a C-terminal inhibitory region and an N-terminal basic domain. The N-terminal region of the protein is necessary and sufficient for targeting of NS to the nucleolus. In its GTP bound state, the inhibitory domain of NS is sequestered, thus freeing the N-terminal region of the protein so it can associate with various nucleolar components. It is through these nucleolar associations that NS accumulates in the nucleolus. GTP exchange promotes association between the N-terminal and inhibitory regions of the protein and NS dissociates from the nucleolus. This novel GTP-driven targeting of NS to the nucleolus is the first defined nucleolar localization mechanism. Our goal is to obtain a detailed structural and biochemical characterization of the GTPase cycle of the protein so we may understand how regulation is occurring in this intriguing protein. This includes determining how NS’s inhibitory region exerts its effects, as well as how the nucleotide bound state of the protein promotes or disrupts specific inter- or intra- domain interactions. Such experiments are necessary to uncover nucleostemin’s role in cell proliferation.